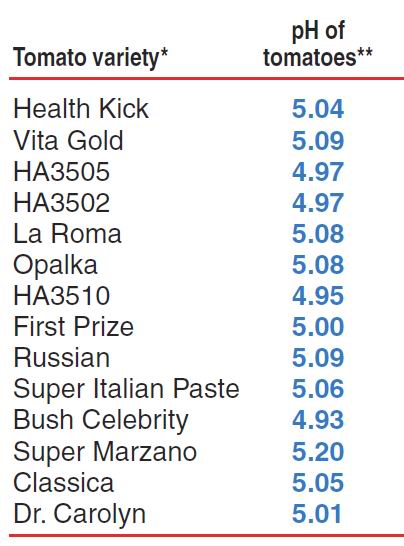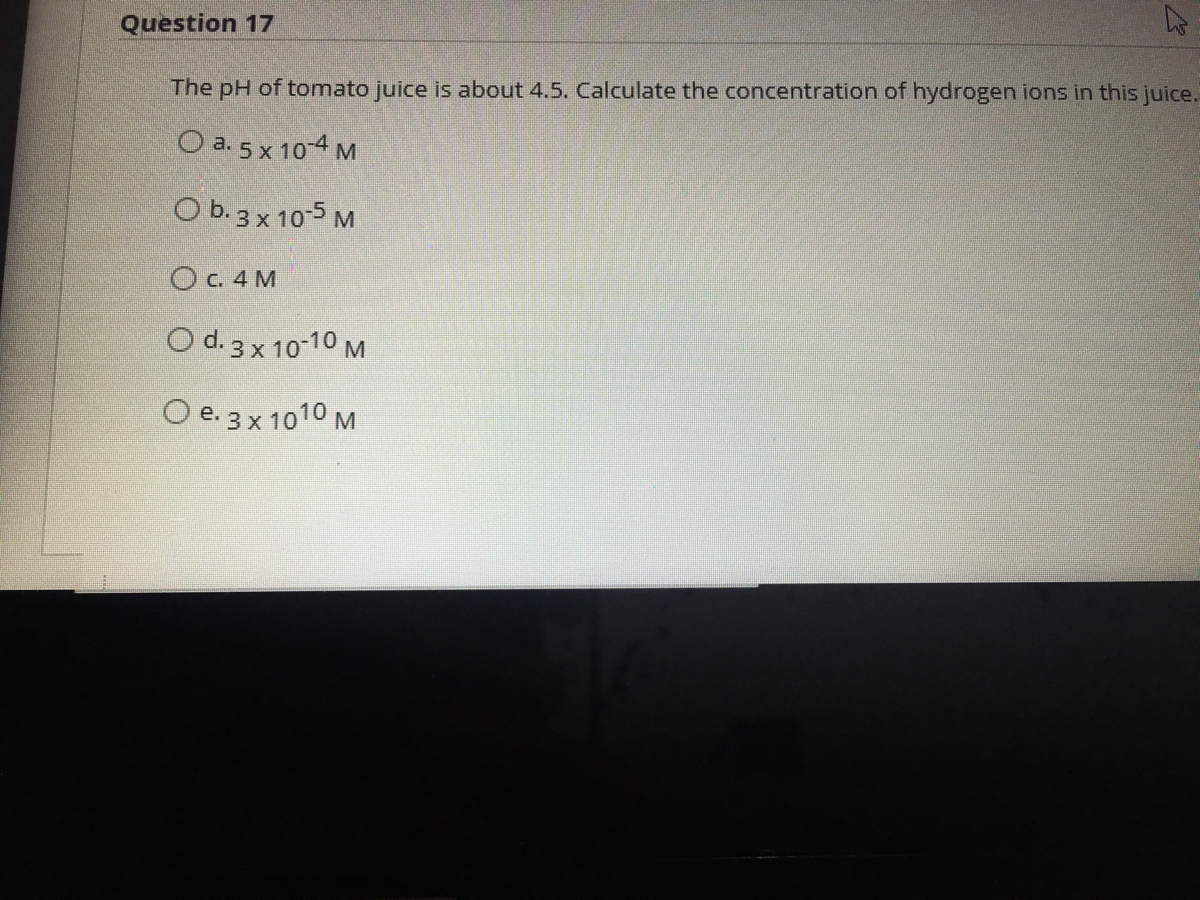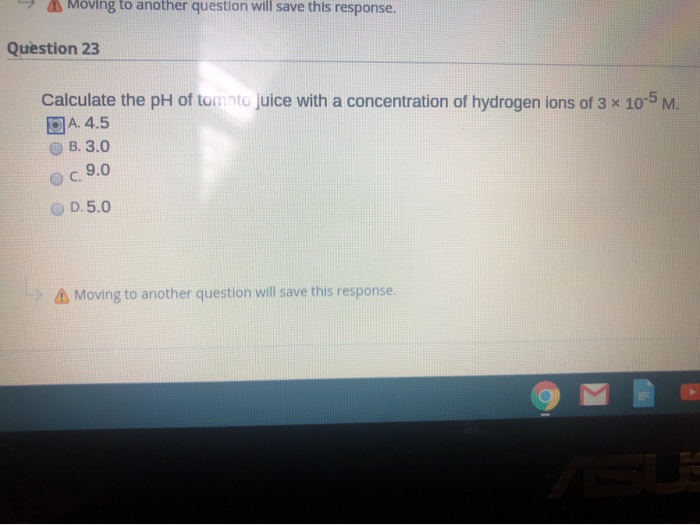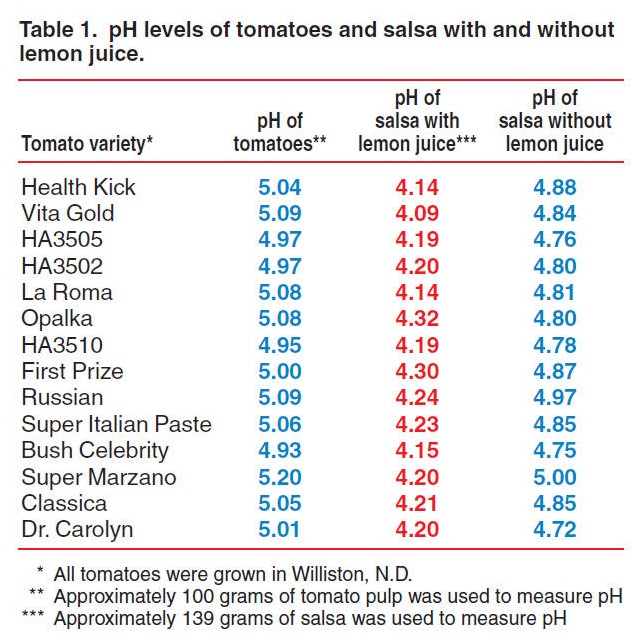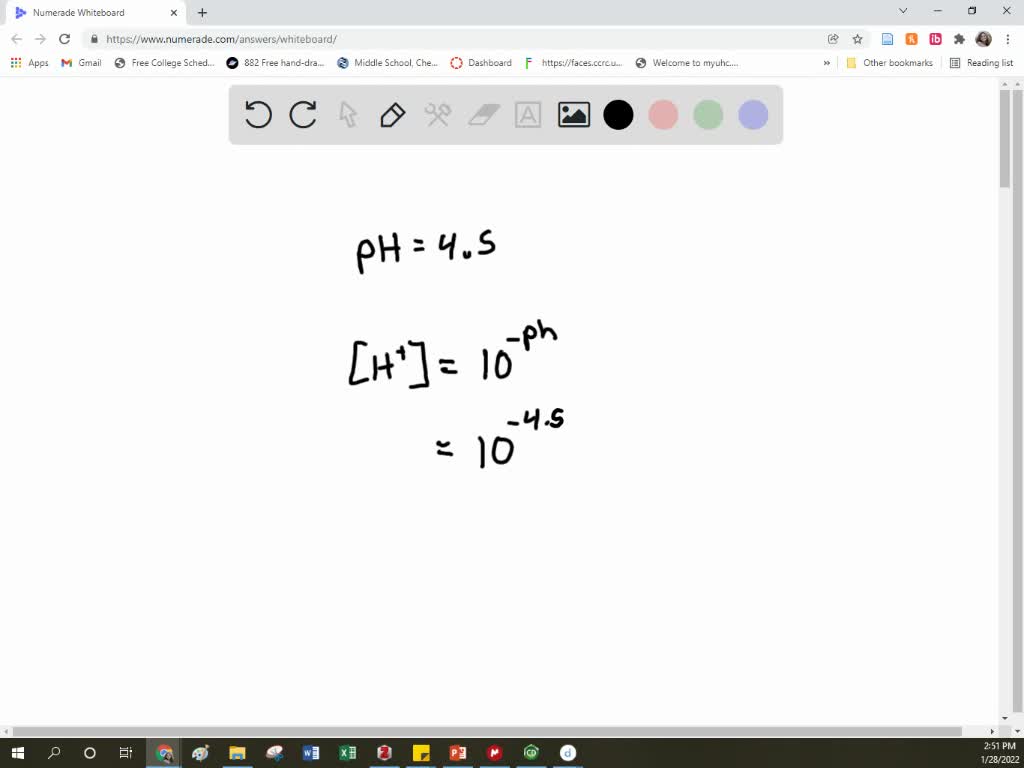
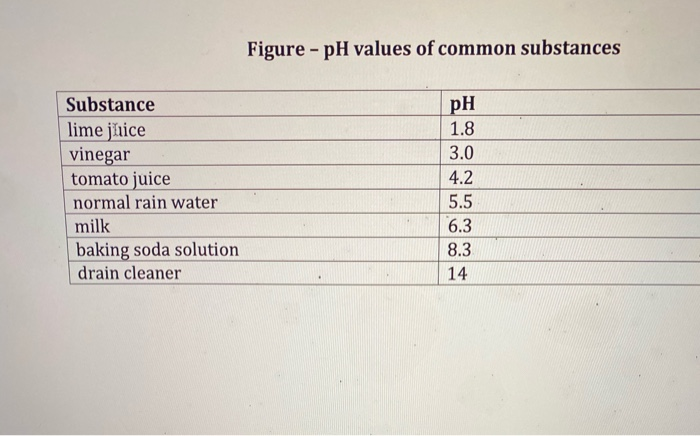
SOLVED: The pH of tomato juice is about 4.5. Calculate the concentration of hydrogen ions in this juice.

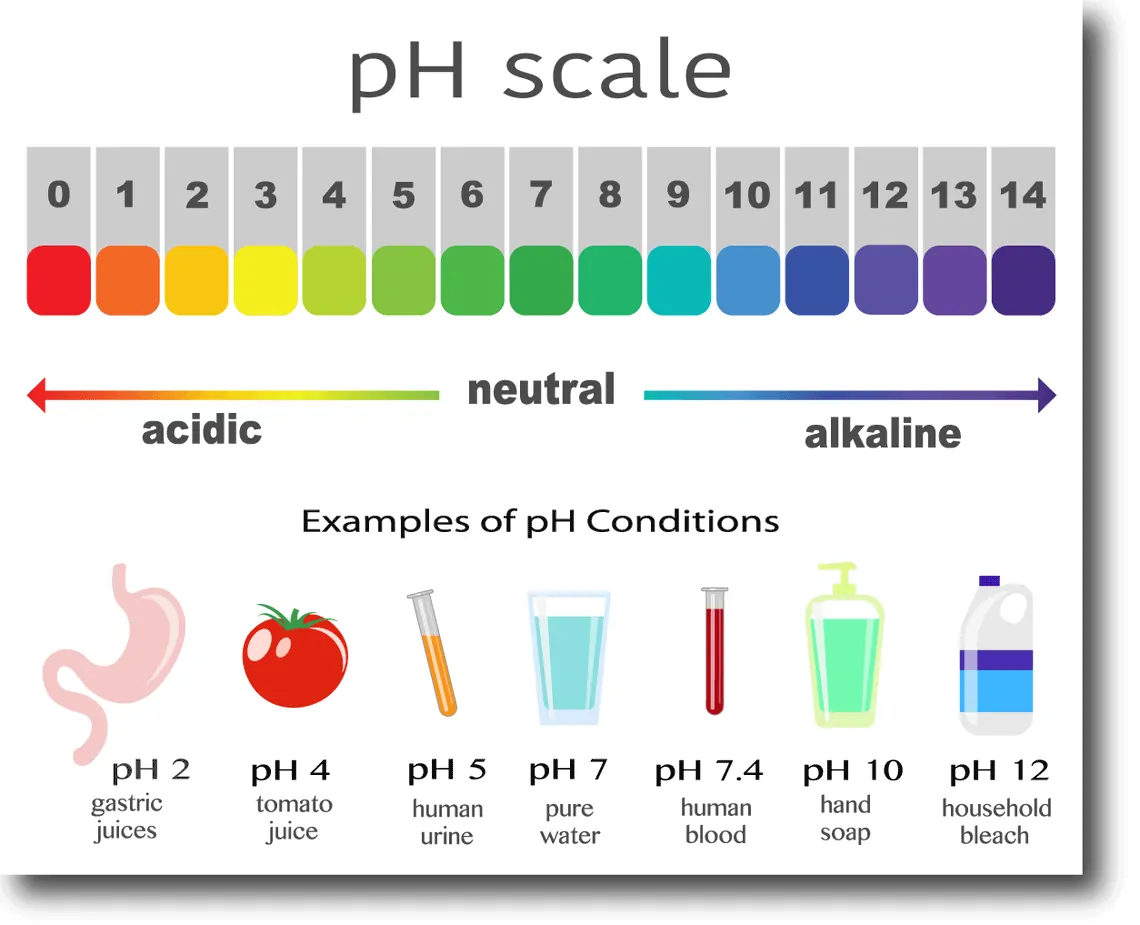
BowTiedGator | Degen Dentist on Twitter: "Chemistry lesson: Lemon juice has a pH of 2. Water pH is 7. pH is logarithmic, which means while it seems like a difference of “5”,
![Learning Check pH4 A. The [H 3 O + ] of tomato juice is 1 x M. What is the pH of the solution? 1) - 42)43)8 B. The [OH - ] of an ammonia solution. - ppt download Learning Check pH4 A. The [H 3 O + ] of tomato juice is 1 x M. What is the pH of the solution? 1) - 42)43)8 B. The [OH - ] of an ammonia solution. - ppt download](https://slideplayer.com/6853055/23/images/slide_1.jpg)











:max_bytes(150000):strip_icc()/how-to-make-tomato-juice-at-home-by-erin-huffstetler-4775102-hero-01-7b0f03dced6e4bf384b1609cf5c965db.jpg)