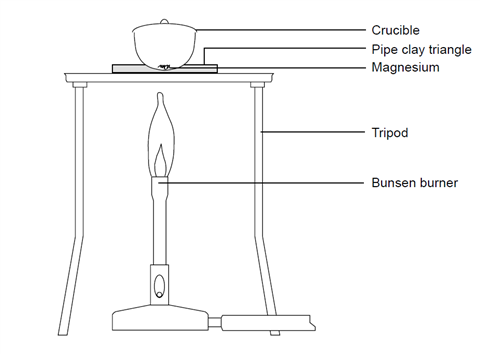
The average atomic mass of a mixture containing 79 mole percent of ^24 Mg and remaining 21 mole percent of ^25 Mg and ^26 Mg , is 24.31. Mole percent of ^26 Mg is :

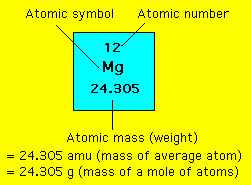
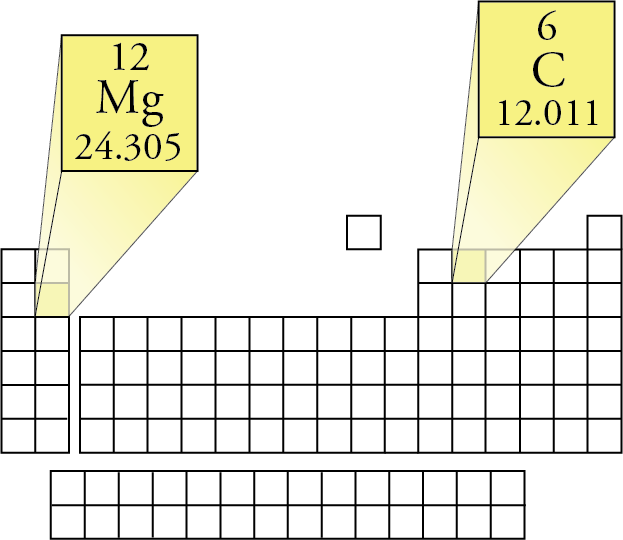
In a periodic table the average atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance on earth. The three isotopes and their

Average atomic mass of magnesium is `24.31`amu. This magnesium is composed of 79 mole % of `24mg... - YouTube
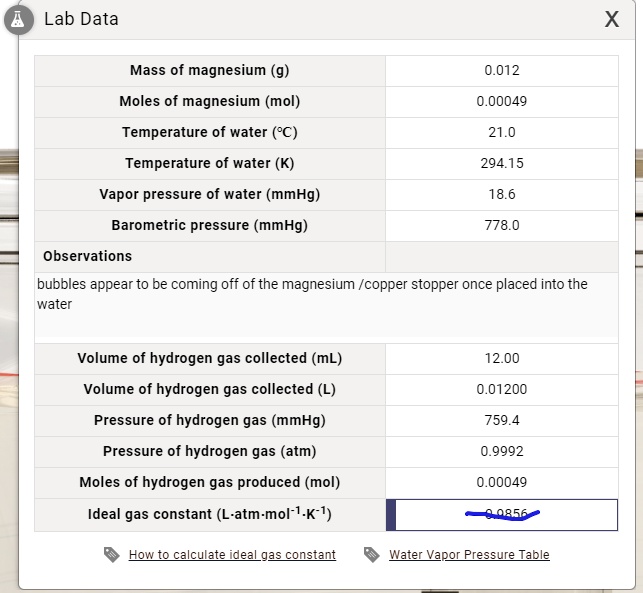
SOLVED: Lab Data X Mass of magnesium (g) 0.012 Moles of magnesium (mol) 0.00049 Temperature of water (PC) 21.0 Temperature of water (K) 294.15 Vapor pressure of water (mmHg) Barometric pressure (mmHg)

View question - Calculate the mass of magnesium oxide formed when 0.52g of magnesium is burned according to the following equation: 2 Mg^s + O^2 ---> 2 MgO^s

2. Using the following data, calculate the average atomic mass of magnesium (give your answer to the - Brainly.com

What mass of magnesium hydroxide is required to neutralize 125 mL of `0.136` M hydrochloric acid - YouTube