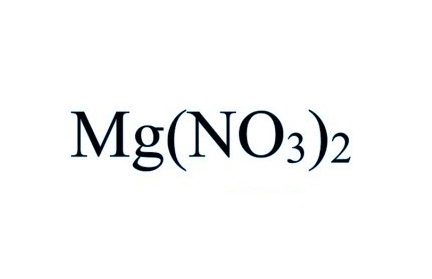A sample of ammonium phosphate, (NH4)3PO4 , contains 3.18 mol of hydrogen atoms. The number of moles of oxygen atoms in the sample is:

Full article: Study on the thermal decomposition mechanism of Mg(NO3)2·6H2O from the perspective of resource utilization of magnesium slag

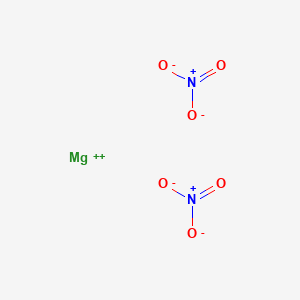
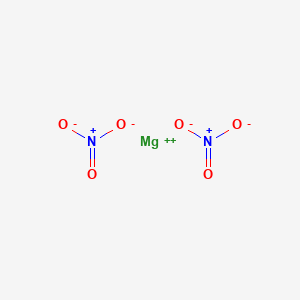
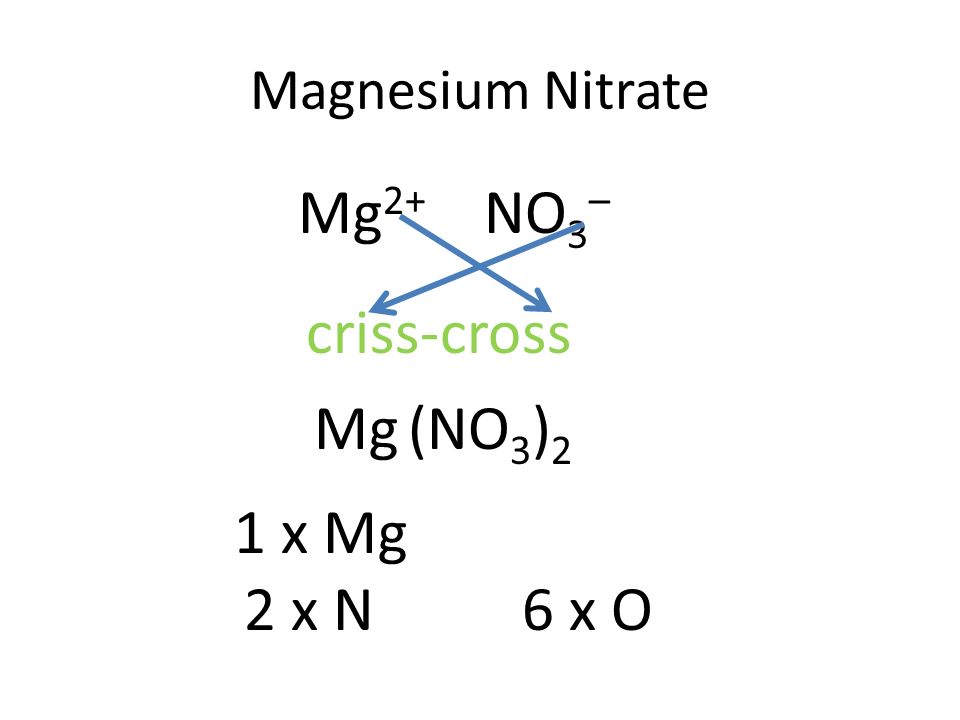
What is the chemical formula of magnesium nitrate Pls answer fast I will send u a - Science - Separation of Substances - 12863787 | Meritnation.com
![Magnesium nitrate] Magnesium nitrate]](https://www.degruyter.com/document/doi/00.0000/IUPAC.iupac.compound.25212/asset/images/25212.png)