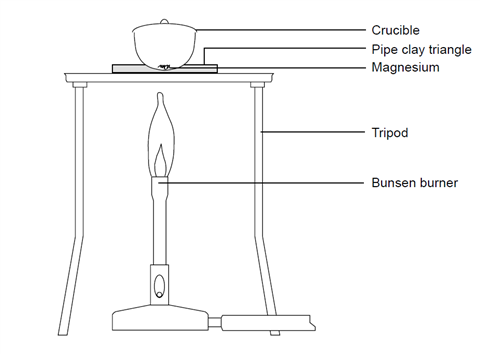
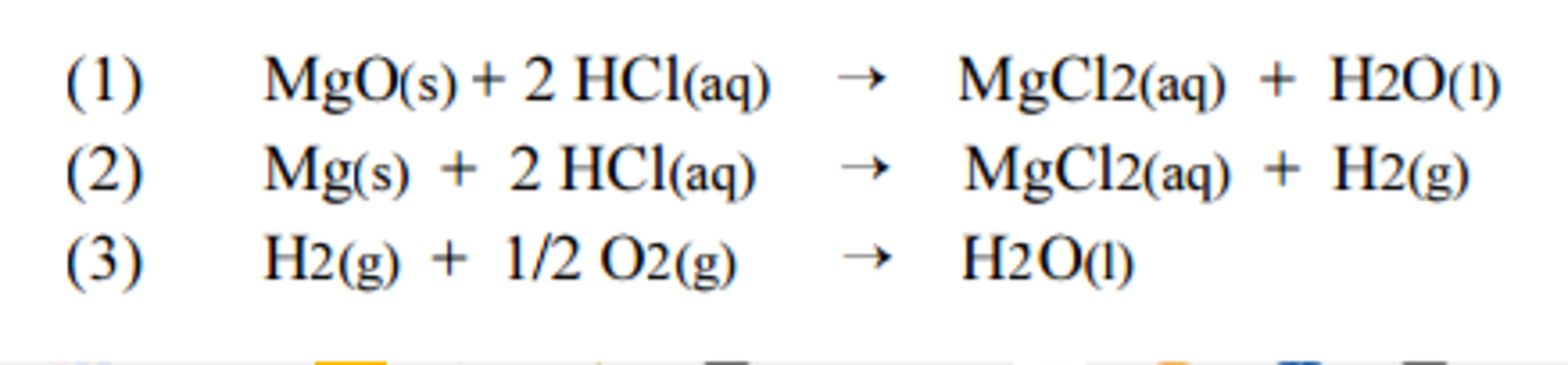Experiment - The Empirical Formula of Magnesium Oxide - International Baccalaureate Chemistry - Marked by Teachers.com

Burning magnesium in a Bunsen flame and other flame experiments | Chem 13 News Magazine | University of Waterloo

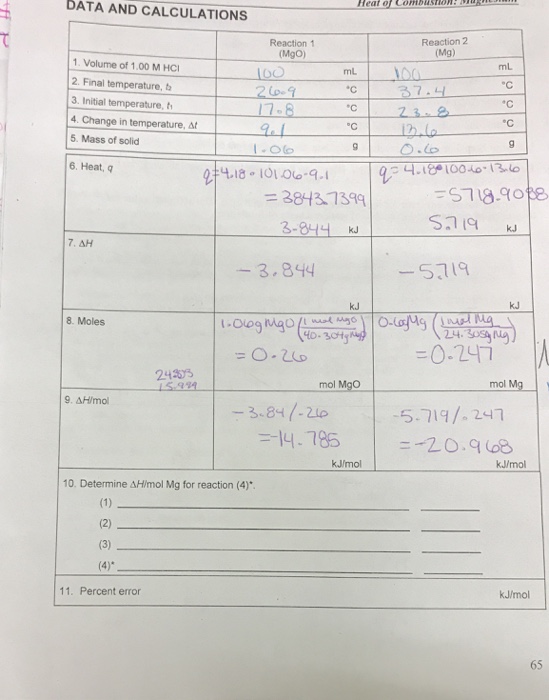
SOLVED: Heat of Combustion of Magnesium In this experiment; we will measure the enthalpy change for the reaction Mg(s) "0 (H) MgO(s) We explored this chemical reaction earlier in the semester when

Hesss Law Lab, use Hesss law to find the enthalpy change of combustion of magnesium which is a highly exothermic reaction. - International Baccalaureate Chemistry - Marked by Teachers.com