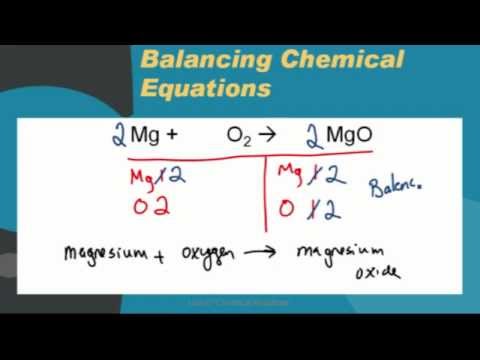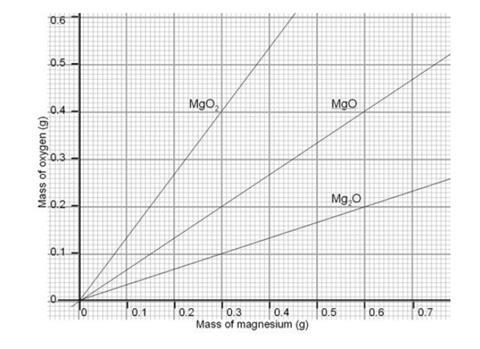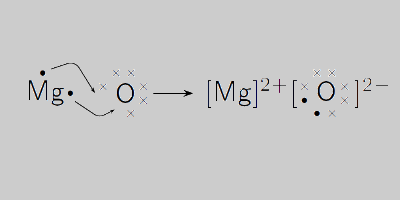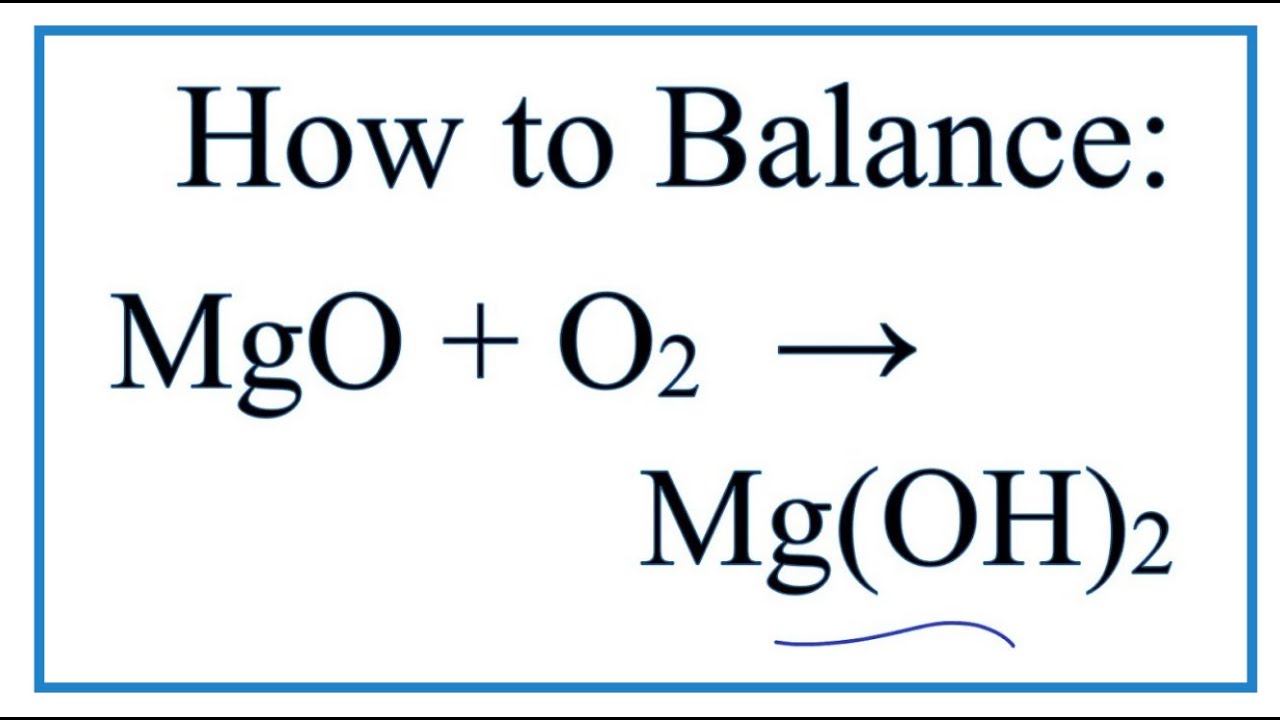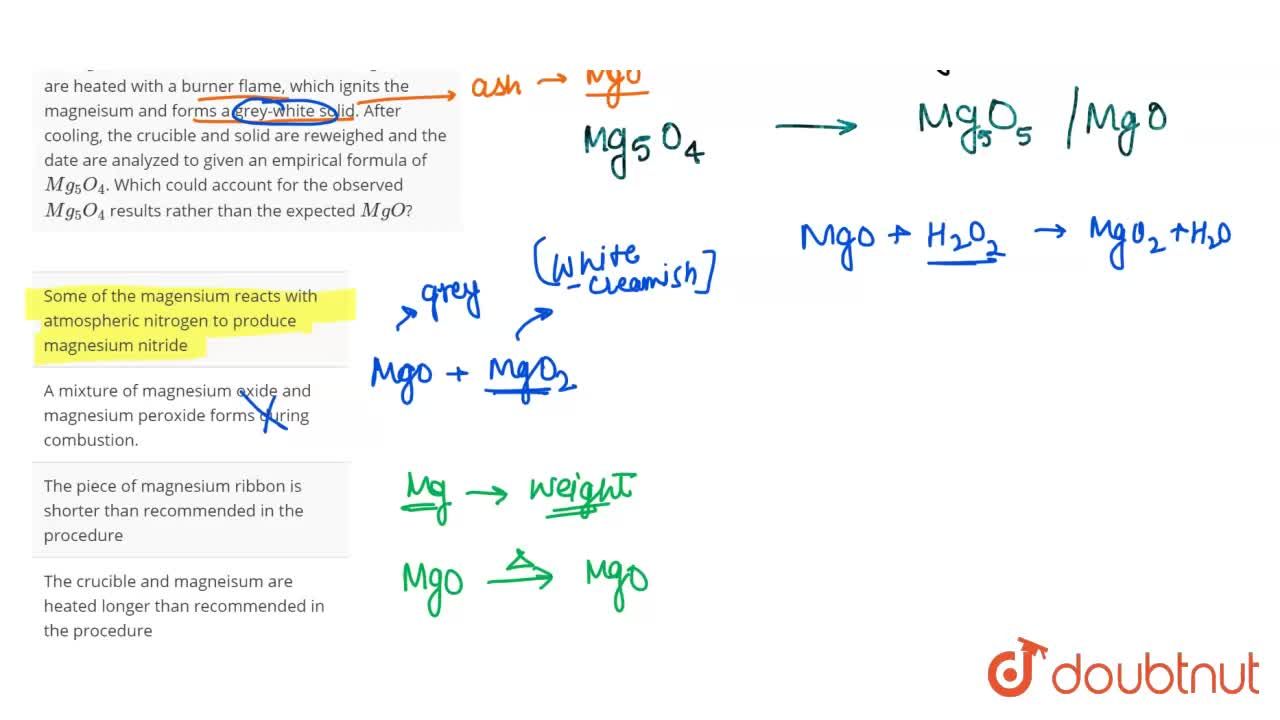
In an experiment to determine the empirical formula of magnesium oxide, a student weights an empty crucible then adds a strip of magnesium metal and reweighs the crucible. The crucible and magnesium are heated with a burner flame, which ignits the ...
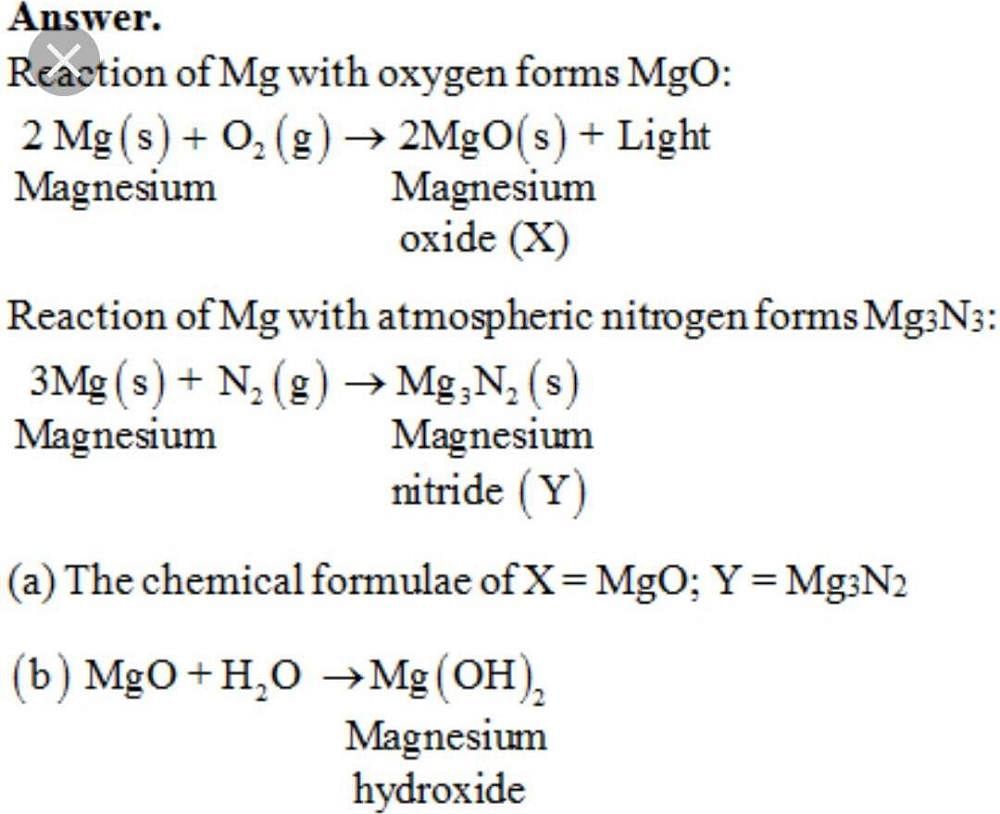
Explain the action of dil.HCL on the following with chemical equations 1- Magnesium ribbon 2-Sodium hydroxide 3-Crushed egg shells? | EduRev Class 10 Question
A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as

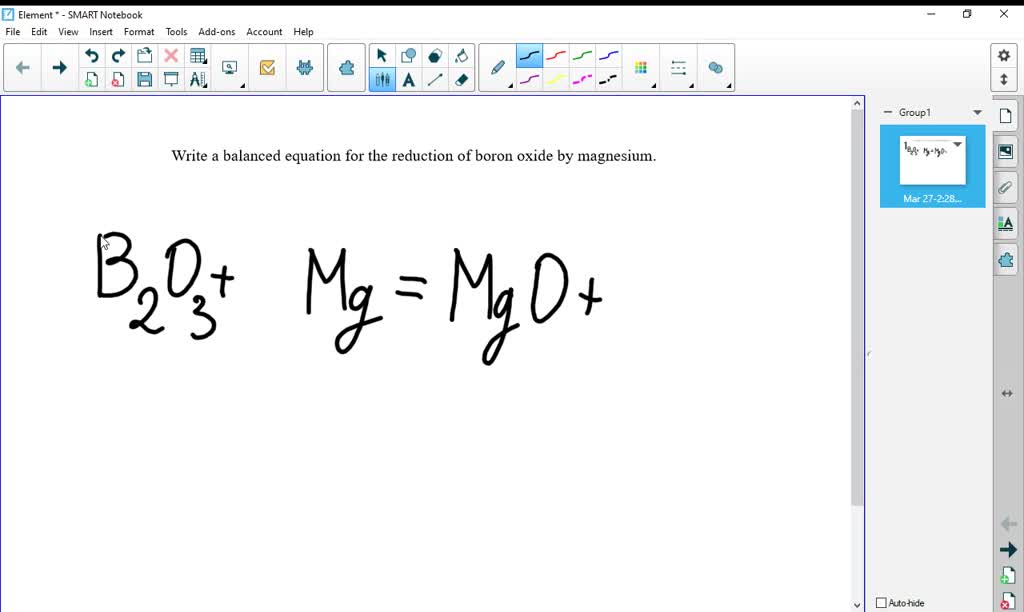
SOLVED: Write the balanced chemical equation for the conversion of magnesium nitride to magnesium oxide as performed in steps 7-8 in the procedure: The reaction produces magnesium oxide and ammonia (NH,). MgzN +
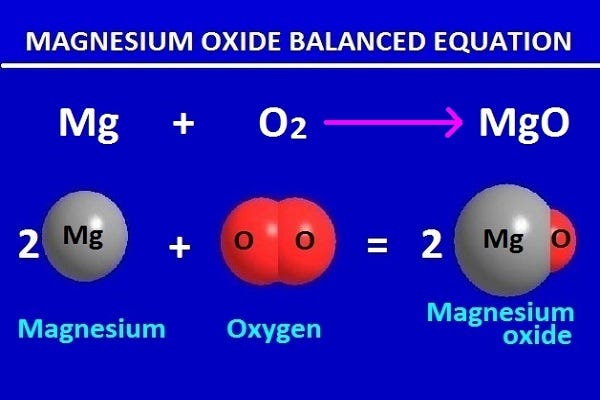
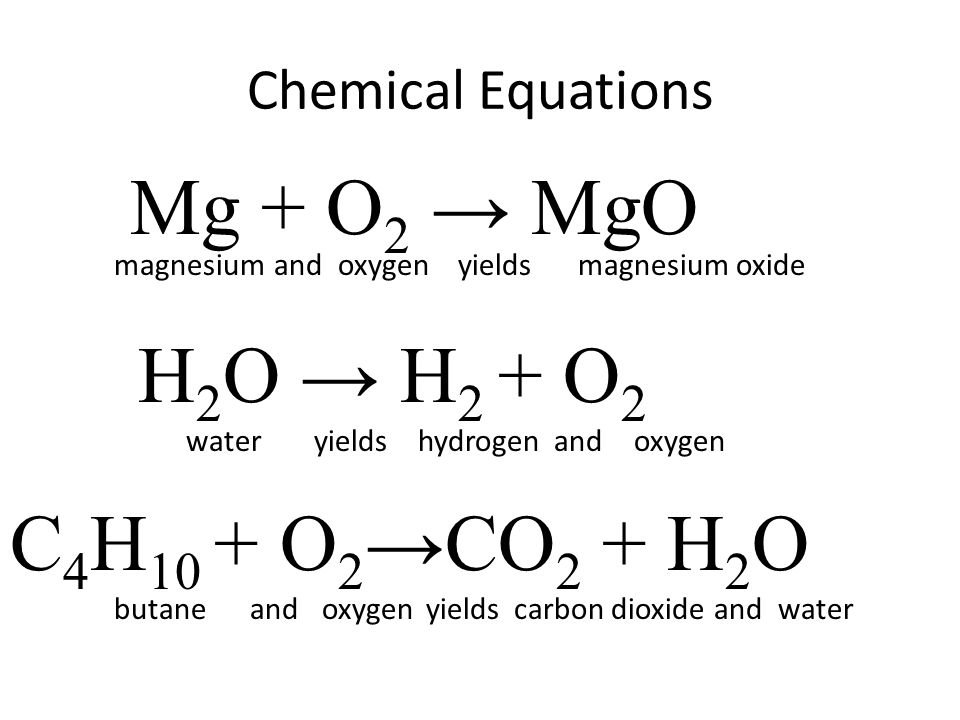
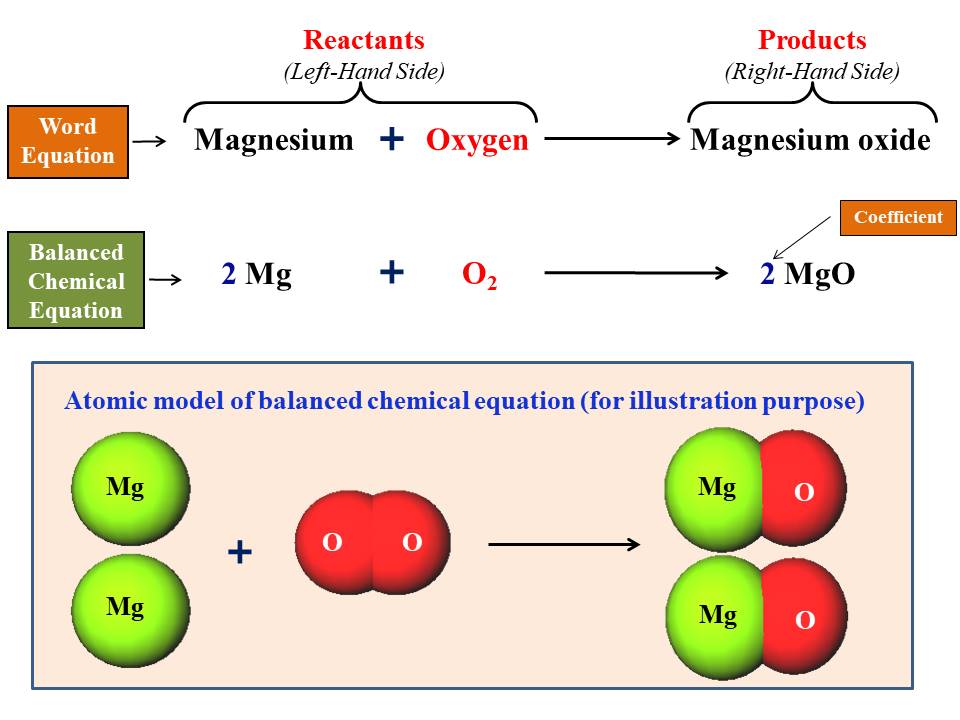
Magnesium oxide balanced equation in chemistry for class 9 | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium